I. 서 론
소화기암이란 위암, 대장암, 직장암, 간암, 담도암, 췌장암 등을 포괄하는 것으로, 인체내에서 음식물을 소화하고 흡수시키기 위한 기본적인 장기에 발생하는 암이다1. 소화기암은 국내암발생률에서 높은 순위를 차지한다. 2014년 국가암등록통계에 따르면, 위암은 국내에서 갑상선암에 이어 두 번째로 많이 발생한 암이며, 대장암은 3위, 간암은 6위, 췌장암은 8위, 담낭 및 기타 담도암은 9위로,대부분 유병률 10위권 이내에 분포하고 있다2.
이렇게 높은 유병률을 나타내는 소화기계암은 근치적 목적뿐아니라 고식적인 효과를 기대하는 경우에도 수술이 일차적 치료방법이 된다3,4. 소화기암 수술이후에는 위장관 운동성저하가 나타날 수 있으며5, 이로 인해 복부팽만감, 장음소실, 장관내 가스축적, 가스배출과 배변지연이 나타난다6. 위장관 운동이상은 마비성 장폐쇄로 발전되기도 하는데, 이는 복부수술의 흔한 합병증이다7. 마비성장폐쇄는 환자의 회복에 악영향을 끼치며, 수술후 사망률을 증가시키고, 입원기간을 연장시키게 된다8.
이러한 수술후 위장관 운동성저하로 인한 마비성 장폐쇄에 한의임상에서는 대건중탕을 많이 사용해왔다. 대건중탕은 후한시대 장중경이 저작한 ≪금궤요략≫에 처음 수록된 처방으로, 촉초, 건강, 인삼, 교이로 구성되어 있다9. 건강과 인삼을 합하여 理中의 의미가 포함되어 있는 방제이다. 촉초의 辛熱은 溫中下氣, 降逆止痛, 殺蟲하며 건강의 辛熱은 溫中祛寒, 痛心助陽, 和胃止嘔, 인삼의 甘溫은 補益脾胃, 大補元氣하여 扶助正氣하며 교이의 甘은建中緩急, 降逆止痛하여 中陽을 건립하고 寒을 거하며, 溫陽을 회복시킨다고 되어있다. 또한 최근에는 장관운동저하, 장협착증, 장이완증, 국한성복막염, 회충복통, 요로결석 등에도 사용되고 있다10.
최근 대건중탕에 대한 실험 및 임상시험은 주로일본에서 많이 이루어지고 있다. 대건중탕이 염증을 억제하고, 장관내 혈류를 증가시키며, 장관운동을 촉진시킨다고 보고된바있다11-13, 또한 지연된 위배출능을 개선시키며14, 상행결장의 배출능을 촉진함을 확인한 연구도 있다15.
대건중탕은 국내 한의임상에서도 많이 사용되고 있지만, 사용근거로 삼을 수 있는 잘 설계된 국내 임상연구는 한편도 없는 실정이다. 90년대초 이후 근거중심의학(evidence based medicine, EBM)의 중요성이 강조되어 왔으나 근거가 될 만한 연구가 많지 않은 상황은 매우 아쉬운 점이다. 이에 저자는 실제 소화기암 수술 후 위장관 운동성 개선을 위해 대건중탕을 사용한 연구들을 분석하여 대건중탕의 효과를 확인하고자 하였다. 또한 기존 연구의 설계를 분석하여, 추후 임상연구설계에도 활용하고자 하였다.
II. 연구방법및절차
1. 문헌검색
최신의 연구동향을 파악하기 위하여 연도는 2000년 이후로 한정하였다. 언어는 한국어와 영어, 중국어로 제한하였다. 해외검색 DB는 Pubmed와 CNKI를 사용하였다. Pubmed에서는 MeSH term인 ‘daikenchuto’(MeSH Unique ID: C121414)를 이용하여 ‘Daikenchuto AND cancer AND ileus’의 검색어로 검색을 시행하였다. CNKI(www.cnki.net)에서는 ‘大建中汤’, ‘肠梗阻’,‘癌’, ‘dajianzhongtang’, ‘ileus’, ‘cancer’의 검색어를 조합하여 사용하였다.
국내 검색 DB로는 한의약 분야와 관련된 데이터베이스인 한국학술정보(http://kiss.kstudy.com), 학술연구정보서비스(http://www.riss.kr), 전통의학정보포털(http://oasis.kiom.re.kr), 한국전통지식포탈(http://www.koreantk.com)을 사용하였고, 검색어로는 ‘암’, ‘대건중탕’을 조합하여 사용하였다. 검색은 2017년 11월 28일에 시행하였다.
2. 문헌분석
문헌의 종류를 분석하였으며, 임상연구인 경우 필수적인 요소인 P(Participants), I(Intervention), C(Comparator), O(Outcome)를 중심으로 분석하였으며, 그 외 임상시험설계에 대해서도 비교하였다.
3. 비뚤림 위험 평가
비뚤림 위험(Risk of Bias) 평가법16에 제시된 7개 항목(무작위 배정순서, 배정 순서 은폐, 연구참여자와 연구자에 대한 눈가림, 결과 평가자 눈가림, 불완전 결과 자료, 선택적 결과 보고, 기타 비뚤림)에 대하여 선정된 연구들을 ‘High risk of bias’, ‘Low risk of bias’, ‘Unclear risk of bias’의 기준에 따라 평가하였다. 2명의 연구자(GJH, SS)에 의해 평가되었으며, 평가자 사이에 의견 불일치가 발생한 경우 재논의를 통해 결정하였다.
III. 결 과
1. 문헌검색결과
Pubmed에서는 27건이 검색되었다. CNKI에서는 ‘大建中汤’ AND ‘肠梗阻’ AND ‘癌’으로 검색한 경우 1편이었다. ‘大建中汤’ AND ‘肠梗阻’으로 일차 검색한 경우에는 24개의 논문이 검색되었으나, 그 중 암수술 이후에 장마비에 대한 논문은 1편이었다. ‘dajianzhongtang’ AND ‘ileus’ AND ‘cancer’로 검색한 결과는 0편이었다. ‘dajianzhongtang’만으로 검색한 결과, 총 3편의 논문이 검색되었으나 암수술 이후에 장마비에 대한 논문은 0건이었다. 국내 DB인 한국학술정보, 학술연구정보서비스, 전통의학정보포털, 한국전통지식포탈에서는 각각0건이 검색되었다. 따라서 총29건의 문헌을 검토하였다. 29편 중 제목과 초록 및 원문을 살펴본 결과, 9개 연구가 최종 포함되었고, 20개 연구를 다음과 같은 이유로 제외하였다.
1) 고찰연구(6편), 2) 비인간대상연구(동물실험, 세포실험)(5편), 3) 방사선 유발 장염에 대한 연구(1편), 4) 수술성 염증반응에 대한 연구(1편), 5) 비임상연구(1편), 6) 복합처방(1편), 7) 원문을 구할 수 없는 문헌(3편), 8) 2000년 이전 연구(2편)
최종 선정 문헌 9편에는 후향적 연구도 포함시켰으며, 임상시험설계를 확인하기 위해 프로토콜 논문도 포함시켰다(Fig. 1).
2. 문헌분석
1) 임상연구 개요 및 설계
9편 모두 일본에서 수행된 연구였으며, 한국에서 수행된 연구는 존재하지 않았다. 9편 중 후향적 연구 1편을 제외하고는 모두 전향적 연구설계였다. 단일기관 임상시험은 3건15,17,20, 다기관 임상시험은 6건이었다4,5,18,19,21,22. 이중맹검을 시행한 논문은 4편이였고4,18,19,21, 무작위 배정을 시행한 연구는 8편4,5,17-22, 교차 설계 연구는 1편이었다22. 위약을 사용한 연구는 5편이었다4,18,19,21,22. 이중임상시험 2상 연구 1편19, 3상 연구도 1편22 존재하였다. 포함된 연구 논문들의 기본적인 특성은 Table 1에 제시하였다(Table 1).
Table 1
The Characteristics of Included Study
| Title | Author, journal, year | Nation | Design | Sample size | Group |
|---|---|---|---|---|---|
| Effect of Daikenchuto (TJ-100) on gastrointestinal symptoms following laparoscopic colectomy in patients with colon cancer: study protocol for a randomized controlled trial17 | Hoshino, Trials,2017 | Japan | single center, randomized, open-label, 2-armed, controlled study | 40 |
A : standard bowel preparation (75 mg sodium picosulfate and 12 mg pursennid) & antibiotic prophylaxis (oral doses of 1 g kanamycin and 750 mg metronidazole) & surgical procedures+Daikenchuto B : standard bowel preparation & antibiotic prophylaxis & surgical procedures |
|
|
|||||
| The effect of Daikenchuto on postoperative intestinal motility in patients with right-side colon cancer15 | Yamada, Surg Today, 2017 | Japan | single center, retrospective, 2-armed, controlled study | 88 |
A : surgery+prophylactic antibiotic flomoxef+Daikenchuto B : surgery |
|
|
|||||
| Evaluation of the efficacy of daikenchuto (TJ-100) for the prevention of paralytic ileus after pancreaticoduodenectomy: A multicenter, double-blind, randomized, placebo-controlled trial21 | Okada, Surgery, 2016 | Japan | multicenter, double-blind, randomized, placebo-controlled trial | 224 |
A : surgery+Daikenchuto (TJ-100) B : surgery+placebo |
|
|
|||||
| Clinical pharmacology of daikenchuto assessed by transit analysis using radiopaque markers in patients with colon cancer undergoing open surgery: a multicenter double-blind randomized placebo-controlled study (JFMC39-0902 additional study)4 | Katsuno, J Gastroenterol, 2016 | Japan | multicenter, double-blind, randomized, placebo-controlled study | 84 |
A : surgery+Daikenchuto B : surgery+placebo |
|
|
|||||
| Effect of Daikenchuto, a Traditional Japanese Herbal Medicine, after Total Gastrectomy for Gastric Cancer: A Multicenter, Randomized, Double-Blind, Placebo-Controlled, Phase Ⅱ Trial19 | Yoshikawa, J Am Coll Surg, 2015 | Japan | multicenter, randomized, double-blind, placebo-controlled, phase ⅱ trial | 207 |
A : surgery+Daikenchuto B : surgery+placebo |
|
|
|||||
| Clinical efficacy of Daikenchuto for gastrointestinal dysfunction following colon surgery: a randomized, double-blind, multicenter, placebo-controlled study (JFMC39-0902)18 | Katsuno, Jpn J Clin Oncol, 2015 | Japan | randomized, double-blind, multicenter, placebo-controlled study | 354 |
A : surgery+Daikenchuto B : surgery+placebo |
|
|
|||||
| Effects of daikenchuto, a Japanese herb, on intestinal motility after total gastrectomy: a prospective randomized trial5 | Akamaru, J Gastrointest Surg, 2015 | Japan | multi-center, open-labeled prospective, randomized, 2-armed trial | 100 |
A : surgery+Daikenchuto B : surgery+placebo |
| Effect of TU-100, a traditional Japanese medicine, administered after hepatic resection in patients with liver cancer: a multi-center, phase Ⅲ trial (JFMC40-1001)22 | Shimada, Int J Clin Oncol, 2015 | Japan | multicenter, phase Ⅲ trial | 231 |
A : surgery+Daikenchuto (TU-100) B : surgery+placebo |
|
|
|||||
| Dai-kenchu-to, a Chinese herbal medicine, improves stasis of patients with total gastrectomy and jejunal pouch interposition20 | Endo, Am J Surg, 2006 | Japan | randomized, cross-over designed study | 17 |
A : initially took Dai-kenchu-to before every meal for 2 weeks (on treatment) and thereafter Daikenchuto was discontinued for 2 weeks (off treatment) B : initially were off treatment for 2 weeks and then were on treatment for 2 weeks |
2) 임상 연구 대상자(Participants)(Table 2)
Table 2
The Inclusion and Exclusion Criteria of the Selected Studies
| Study | Inclusion criteria | Exclusion criteria |
|---|---|---|
| Hoshino, 201717 |
1. Patients with left-sided colon cancer (including rectosigmoid cancer) who are scheduled to undergo laparoscopic surgery 2. Clinical stage I, II and III 3. Patients who suffer from abdominal pain and distention at POD 1 (NRS score≥1) 4. The European Cooperative Oncology Group (ECOG) performance status of 2 or less 5. Patients aged 20 years and older at registration 6. Patients who can take medications orally 7. Written informed consent provided to participate in the study |
1. Patients who have history of abdominal surgery or history of bowel obstruction 2. Patients with concomitant inflammatory bowel disease such as ulcerative colitis and Crohn’s disease 3. Patients with concomitant endometriosis 4. Patients requiring emergency surgery 5. Patients who have been or will be treated by chemotherapy or radiotherapy 6. Patients with severe comorbidity such as cardiac disease, liver disease, pulmonary disease or renal disease 7. Patients who took Japanese herbal medicine (Kampo) up to 4 weeks prior to registration 8. Patients who took gastrointestinal prokinetic drugs, antipsychotic drugs or antidepressant drugs up to 4 weeks prior to registration 9. Patients with a history of a Kampo allergy in other formulations 10. Patients with hepatitis B or C 11. Patients who are unable to take medications orally at POD 1 12. Patients who are unsuitable for study inclusion as determined by the nvestigator (e.g., those with severe dementia) |
|
|
||
| Yamada, 201715 |
1. With primary tumors located in the right-side colon 2. Who could undergo elective surgery 3. Who had American Society of Anesthesiologists grades of I-Ⅲ 4. Who were aged 20-85 years |
1. ≥2 anastomoses 2. Intestinal obstruction before surgery (peristaltic activity is impaired in this condition) 3. Having undergone colostomy or ileostomy 4. Having undergone any additional procedure 5. Requiring admission to the intensive-care unit after surgery. |
|
|
||
| Okada, 201621 | 1. Adults (≥20 years of age) who were scheduled to undergo pancreaticoduodenectomy for periampullary tumors and tumors of the head of the pancreas |
1. Patients with tumors that required resection of the colon 2. Patients who were expected to have severe intra-abdominal adhesions as the result of previous surgery or peritonitis 3. Patients who had used gastrointestinal prokinetic medication, antipsychotic medication, or antidepressants 4. Patients who had used Japanese herbal (Kampo) medicines within 4 weeks before registration |
|
|
||
| Katsuno, 20164 |
1. Qualified for curative open surgery for sigmoid cancer (including cancer of the rectosigmoid region) on the basis of preoperative diagnosis of stage I, Ⅱ, Ⅲa, or Ⅲb disease and T category of 1-3, N category of 0-2, and M category of 0 2. Performance status of 0-1 3. Able to tolerate oral administration of Daikenchuto 4. 20 years or older 5. Able to stay in the hospital during the study period 6. Able to provide written informed consent. |
1. Scheduled for endoscopic surgery 2. Scheduled for laparoscopic surgery 3. Had complicated inflammatory bowel disease (ulcerative colitis or Crohn’s disease) 4. Required emergency surgery 5. Had double cancer 6. Had a serious liver disorder 7. Had a serious renal disorder 8. Had a history of laparotomy and peritonitis (except surgery for appendicitis) 9. Taking other Kampo medicine 10. Pregnant, possibly pregnant, lactating, or considering a pregnancy 11. Unfit for the study as determined by the attending physician |
|
|
||
| Yoshikawa, 201519 |
1. Gastric cancer and were planning open total gastrectomy with Roux-en-Y reconstruction 2. Had an Eastern Cooperative Oncology Group performance status of 0 to 1 3. Were capable of orally taking test reagents 4. Were aged from 20 to 85 years 5. Had sufficient function of vital organs (including bone marrow, heart, liver, kidneys, and lungs) 6. Were in an adequate general condition to undergo total gastrectomy 7. Were inpatients during the study period 8. Provided written informed consent |
1. Previous laparotomy (except appendectomy) 2. Previous intestinal resection 3. Ulcerative colitis or Crohn’s disease 4. Emergency operation 5. A diagnosis of cancer before the current gastric carcinoma 6. Chemotherapy in the 4 weeks before surgery or during the trial period 7. Intake of other Kampo medicines in the 4 weeks before surgery 8. Patients who were pregnant or possibly pregnant and those who had synchronous cancers |
|
|
||
| Katsuno, 201518 |
1. Qualified for curative colonic open resection for colon cancer (including cancer of the rectosigmoid) that had been diagnosed pre-operatively according to the disease staging (I, Ⅱ, Ⅲa, Ⅲb, TNM category distribution: T=1-3, N=0-2, M=0) 2. Diagnosed with a performance status (PS) of 0-1 3. Able to tolerate oral administration of DKT 4. Aged 20 years or older 5. Man or woman 6. Able to stay in hospital during the entire length of study period; and 7. Able to provide written informed consent |
1. Those scheduled for endoscopic or laparoscopic surgery 2. Those having complicated inflammatory bowel disease (ulcerative colitis and Crohn’s disease) 3. Those requiring emergency surgery 4. Those diagnosed with double cancer, serious liver disorder or serious renal disorder 5. Those with a history of laparotomy and peritonitis (excluding surgery for appendicitis) 6. Those taking other Kampo medicines 7. Those who were pregnant, possibly pregnant, lactating or considering pregnancy 8. Those unfit for the study as determined by the attending physician |
|
|
||
| Akamaru, 20155 |
1. Histologically confirmed stage I, Ⅱ, or Ⅲ gastric cancer with a plan to undergo total gastrectomy with a D2 dissection (permitting preservation of the spleen), Roux-en-Y reconstruction, and R0 surgery 2. No previous cancer treatment or past history of any other cancer 3. Age between 20 to 80 years 4. An Eastern Cooperative Oncology Group performance status of 0 or 1 5. Adequate organ functions |
1. Any hepatic, peritoneal, or distant metastasis or any positive tumor cells in cytological examinations of peritoneal fluids 2. Emergency surgery, other active malignancies, morbid cardiopulmonary disease, severe liver-kidney dysfunction, a history of laparotomy (except appendectomy), or intestinal obstruction. |
|
|
||
| Shimada, 201522 |
1. Were planning open or laparoscopy-assisted hepatic resection of laparotomy 2. Had an Eastern Cooperative Oncology Group performance status of 0-2 3. Were capable of orally taking test reagents 4. Were aged over 20 5. Had no history of prior chemotherapy or radiotherapy within 4 weeks before surgery 6. Had sufficient vital organ functions (bone marrow, heart, liver, kidney, lung, etc.) 7. Were in a satisfactory general condition for hepatectomy 8. Showed serum C-reactive protein (CRP) levels of 2.0 mg per deciliter 9. Were inpatients during the study period 10. Provided their written informed consent. |
1. Planning a pure laparoscopic hepatectomy 2. A history of hepatectomy 3. Comorbidity of ulcerative colitis or Crohn’s disease 4. Emergency operation(s) 5. Synchronous and metachronous gastrectomy 6. Synchronous colectomy combined treatment with radiofrequency ablation 7. Prior chemotherapy within 4 weeks or intraoperative chemotherapy or radiation therapy 8. Planned chemotherapy or radiotherapy within 10 days after surgery 9. Intake of other Kampo medicines within 4 weeks before surgery 10. Pregnancy or possible pregnancy 11. Reconstruction of biliary tract 12. Colostomy 13. Simultaneous administration of synbiotics 14. Patients judged inappropriate for this study by the physicians |
|
|
||
| Endo, 200620 | 1. Who underwent total gastrectomy with jejunal pouch interposition for gastric carcinoma |
1. No significant associated diseases such as cirrhosis, renal failure, pulmonary diseases, or cardiac diseases. 2. Not had a recurrence or any postoperative complications including leakage or intestinal obstruction |
(1) 연령의 경우 하한이 20세 이상이었고, 상한은 85세였다. 상한 혹은 하한이 제시되지 않은 경우도 있었다. 연구대상자수는 17명에서 254명까지 다양했다.
(2) 임상 연구 대상자별로 살펴보면, 대장암으로 인한 대장절제환자를 대상으로 한 논문 4편4,16-18, 위암으로 인한 위전절제환자대상 논문 3편5,19,20, 췌장암으로 인한 췌십이지장절제환자 대상 논문 1편21, 간암으로 인한 간절제환자 대상 논문 1편으로 분류되었다22.
(3) 선정기준
암의 병기측면에서는 1-3기 모두를 대상으로 한 연구가 5편이었으며4,5,15,17,18, 특별히 병기에 대해 언급하지 않은 논문이 4편이었다. 또한 환자 상태에 대해 Eastern Cooperative Oncology Group performance status(ECOG)를 적용한 논문은 8편이었으며4,5,15,17-19,21,22, 이중 5편은 ECOG 0에서 1사이의 환자를 대상으로 하였고, 2편은 0에서 2사이의 환자를 대상으로 하였다. 복강경수술을 시행한 연구는 2편이었으며17,22, 3편의 연구는 개복술로 진행하였고4,18,19, 다른 연구는 복강경, 개복술 여부에 대한 특별한 기술은 없었다. 서면동의서를 받은 자를 선정기준에 포함시킨 논문은 5편이었다4,17-19,22. 한편, 특이한 선정기준을 제시한 몇 개의 연구가 있었는데 Endo의 연구에서는 위전절제를 시행하면서 공장주머니삽입술을 시행한 자를 대상으로 하였고20, Shimada의 연구에서는 C-reactive protein 수치가 2.0 mg/dL 이하인 대상자를 모집하였다22. Yamada의 연구에서는 선정기준에 American Society of Anesthesiologists grades를 포함시켰다15. 자세한 선정기준은 Table 2에 제시하였다(Table 2).
(4) 제외기준
3) 시험약(Intervention)(Table 3)
Table 3
The Characteristics of Intervention in the Included Studies
| Study | Intervention | Contents & dose & period |
|---|---|---|
| Hoshino, 201717 | Daikenchuto |
Daikenchuto (5 g) will be administered orally three times per day between POD 2 and POD 28. DKT manufactured by Tsumura & Co (Tokyo, Japan) will be used. |
|
|
||
| Yamada, 201715 | Daikenchuto | 7.5 g of Daikenchuto was administered for five days starting the second day after surgery. |
|
|
||
| Okada, 201621 | Daikenchuto | A solution of 5 g of TJ-100 was administered orally 3 times daily (15 g/day) immediately before meals or every 8 hours for 17 consecutive days (from preoperative day 3 to postoperative day 14), except on the operative day and on postoperative day 1; on the operative day, TJ-100 was administered immediately after the operation in a single 5 g dose as a diluent via a delivering tube (10 Fr) inserted into jejunum; and on postoperative day 1, 3 doses of 5 g were delivered via a delivering tube to prevent aspiration pneumonia. TJ-100 extract powder is manufactured by Tsumura & Co. (Tokyo, Japan) |
|
|
||
| Katsuno, 20164 | Daikenchuto |
Daikenchuto orally at a dosage of 15 g/day (5 g three times a day) from postoperative day 2 to postoperative day 8. Daikenchuto was manufactured by Tsumura & Co. (Tokyo, Japan) |
|
|
||
| Yoshikawa, 201519 | Daikenchuto |
Oral doses of 15 g/d (5 g 3 times a day) of either of Daikenchuto from postoperative day (POD) 1 or 2 to 12. Daikenchuto was administered by oral or nasogastric tube. Daikenchuto were manufactured by Tsumura & Co. |
|
|
||
| Katsuno, 201518 | Daikenchuto | Oral doses of 15 g/day (5 g t.i.d) of Daikenchuto from post-operative day (POD) 2 to POD8. Daikenchuto were manufactured by Tsumura & Co. (Tokyo, Japan). |
|
|
||
| Akamaru, 20155 | Daikenchuto | 2.5 g of Daikenchuto (Tsumura & Co., Tokyo, Japan), taken orally with 20 ml tepid water three times per day, starting the day after the operation, when oral intake was allowed. They continued treatment through the third month after surgery. |
|
|
||
| Shimada, 201522 | Daikenchuto | Oral doses of 15 g/day (5 g three times a day) of TU-100 from preoperative day 3 to postoperative day 10, except on the day of liver surgery. TU-100 was manufactured by Tsumura & Co. (Tokyo, Japan) |
|
|
||
| Endo, 200620 | Daikenchuto | Initially took 15 g/d of Daikenchuto (Tsumura & Co., Tokyo, Japan) before every meal for 2 weeks (on treatment) and thereafter Daikenchuto was discontinued for 2 weeks (off treatment) |
4) 대조군(Comparator)(Table 4)
Table 4
Characteristics of Comparator in the Included Studies
| Study | Comparator |
|---|---|
| Hoshino, 201717 | No additional medicine will be administered as a comparator. |
|
|
|
| Yamada, 201715 | No additional medicine was administered as a comparator. |
|
|
|
| Okada, 201621 | Daikenchuto placebo: a solution of 5 g of placebo was administered orally 3 times daily (15 g/day) immediately before meals or every 8 hours for 17 consecutive days (from preoperative day 3 to postoperative day 14), except on the operative day and on postoperative day 1; on the operative day, placebo was administered immediately after the operation in a single 5 g dose as a diluent via a delivering tube (10 Fr) inserted into jejunum; and on postoperative day 1, 3 doses of 5 g were delivered via a delivering tube to prevent aspiration pneumonia. |
|
|
|
| Katsuno, 20164 | Daikenchuto placebo: placebo orally at a dosage of 15 g/day (5 g three times a day) from postoperative day 2 to postoperative day 8 |
|
|
|
| Yoshikawa, 201519 |
Daikenchuto placebo: oral doses of 15 g/d (5 g 3 times a day) of either of DKT placebo from postoperative day (POD) 1 or 2 to 12. Daikenchuto placebo was administered by oral or nasogastric tube. |
|
|
|
| Katsuno, 201518 | Daikenchuto placebo: oral doses of 15 g/day (5 g t.i.d) of DKT placebo from post-operative day (POD) 2 to POD8. DKT placebo were manufactured by Tsumura & Co. (Tokyo, Japan). |
|
|
|
| Akamaru, 20155 | 20 ml tepid water three times per day |
| Shimada, 201522 | TU-100 placebo: oral doses of 15 g/day (5 g three times a day) of TU-100 placebo from preoperative day 3 to postoperative day 10, except on the day of liver surgery |
|
|
|
| Endo, 200620 | Initially were off treatment for 2 weeks and then were on treatment (15 g/d of Daikenchuto before every meal for 2 weeks) for 2 weeks. |
5) 유효성 평가지표(Outcome)(Table 5)
Table 5
The Characteristics of Outcomes in the Included Studies
| Study | Outcome |
|---|---|
| Hoshino, 201717 |
1. Primary endpoints 1) Grade of abdominal pain determined using the numeric rating scale (NRS) 2) Grade of abdominal distention determined using the NRS 3) Quality of life determined using the Gastrointestinal Quality Life Index (GIQLI) NRS measurements of abdominal pain and distention will be taken prior to surgery and at postoperative days (POD) 1, 4, 7, 14 and 28. GIQLI will be taken before surgery and at POD 14 and 28. 2. Secondary endpoints 1) Postoperative nutritional status (Onodera’s prognostic nutritional index (PNI) & Controlling Nutritional Status score (CONUT score) 2) Time to initial flatus 3) Time to initial defecation 4) Bowel gas volume measured using analysis software 5) Character of stool (Bristol Stool Form Scale) 6) Defecation frequency per day 7) Postoperative complications (Clavien-Dindo classification) 8) Duration of postoperative hospital stay 9) Metabolomics analysis of metabolites in stool and blood using gas chromatography-tandem mass spectrometry (GC/MS/MS) and liquid chromatography-tandem mass spectrometry (LC/MS/MS) |
|
|
|
| Yamada, 201715 |
1. Primary endpoints: the effect of Daikenchuto on the postoperative intestinal motility assessed by the radiopaque marker Sitzmarks (Konsyl, Easton, MD, USA), which contains 20 radiopaque ring markers (diameter: 4.5 mm) per capsule.) The postoperative intestinal motility was assessed radiologically by counting the number of residual markers on abdominal radiography (PODs 1, 3, and 5). 2. Secondary endpoint:the postoperative inflammation reaction by C-reactive protein (CRP) on POD 3 |
|
|
|
| Okada, 201621 |
1. Primary endpoints 1) Incidence of postoperative paralytic ileus lasting >72 hours after surgery 2) Time to occurrence of postoperative paralytic ileus 2. Secondary endpoints 1) The incidence of postoperative paralytic ileus in cases which combined with/without enteral alimentation 2) Quality of life (QOL) assessment by the Gastrointestinal Symptom Rating Scale Score (Japanese version) 3) Assessment of abdominal pain and abdominal distention by means of a visual analog scale 4) The ratio of the abdominal circumference on postoperative day 3 to the circumference on the operative day immediately after surgery 5) The incidence of postoperative complications, |
|
|
|
| Katsuno, 20164 |
1. Total transit analysis (the total number of evacuated radiopaque markers 72 h after administration) 2. Segmental transit analysis at 6, 24, and 72 h (the number of markers identified on radiographs in the following locations was determined by three physicians: 1) in the anal side of the small intestine at 6 h (evacuation from the stomach), 2) the anal side of the cecum at 24 h (evacuation from the small intestine), and 3) the anal side of the sigmoid colon at 72 h.), 3. The time to first flatus |
|
|
|
| Yoshikawa, 201519 |
1. Primary endpoints: time from the end of operation (tracheal tube extubation) until first flatus and defecation, and frequency of defecation per day after surgery. 2. secondary endpoints 1) Quality of life according to the Gastrointestinal Symptom Rating Scale (Japanese Version) and Functional Assessment of Cancer Therapy-Gastric FACT-Ga 2) Serum C-reactive protein levels (3) Presence or absence of severe postoperative bowel movement disorder (4) Presence or absence of postoperative ileus (5) Adverse events (National Cancer Institute Common Terminology Criteria for Adverse Events) |
|
|
|
| Katsuno, 201518 |
1. Primary endpoints: the time from the end of surgery (tracheal tube extubation) until first bowel movement, frequency of bowel movement per day and change in the Bristol Stool Scale (BSS) scores after surgery 2. Secondary endpoints 1) Quality of life (QOL) according to the Gastrointestinal Symptom Rating Scale (GSRS, Japanese version) and Functional Assessment of Cancer Therapy-Colorectal (FACT-C) scale 2) Serum C-reactive protein (CRP) levels 3) The incidence of intestinal obstruction 4) Adverse events (AEs) (National Cancer Institute Common Terminology Criteria for Adverse Events). |
|
|
|
| Akamaru, 20155 |
1. Gut motor functions during the hospital stay (time to first bowel movement and the frequency and properties of stools) 2. A radiographic quantification of bowel gas 3. QOL assessment 4. The incidence of postoperative intestinal obstruction 5. Adverse events related to the Daikenchuto medicine |
|
|
|
| Shimada, 201522 |
1. Primary endpoints: the time from extubation until the first postoperative bowel movement (FBMT), serum CRP levels (days -1, 1, 3, 5, 7, 10), serum ammonia levels (days -1, 1, 3, 5, 7, 10) 2. Secondary endpoints 1) The presence or absence of postoperative ileus or other complications 2) Postoperative hospital stay 3) Adverse events (National Cancer Institute Common Terminology Criteria for Adverse Events) |
|
|
|
| Endo, 200620 |
1. Postprandial symptoms caused by stasis including upperabdominal fullness, upper-abdominal discomfort, upper-abdominal pain, regurgitation, and lack of hunger using the Visick grading scale with modifications 2. A dual-phase scintigraphic study 3. Manometric study |
(1) 수술후 위장관 기능 평가
다음과 같은 다양한 평가지표들이 사용되었다: ▲수술후 첫 배변까지의 시간17 ▲수술후 첫 가스배출까지의 시간4,17,19 ▲수술후 첫 장관운동까지의 시간5,18,22 ▲1일 배변횟수5,17,19 ▲배변양상17,18 ▲1일 장운동횟수18 ▲수술후 마비성장폐색증 발생률5,18,21 및 발생여부19,22 ▲수술후 마비성장폐색증 발생까지의 시간21 ▲수술후 심각한 장관운동이상 존재 여부19 ▲수술후 장관운동성 평가-radiopaque marker4,15, manometric study20 ▲위장관배출능 평가- scintigraphic study20 ▲장관내 가스양5
(2) 삶의 질
삶의 질은 4편의 연구에서 평가되었다. 삶의 질 측정도구는 다양했는데 Gastrointestinal Symptom Rating Scale(GSRS)가 가장 많았고5,18,19,21, 그 외에 Gastrointestinal Quality Life Index(GIQLI)17, Functional Assessment of Cancer Therapy-Gastric FACT-Ga19 (1편), Functional Assessment of Cancer Therapy- Colorectal(FACT-C)18 등이 GSRS와 함께 사용되었다.
(3) 혈액학적 검사
(4) 증상점수
Hoshino의 연구에서는 복부통증과 복부팽만감의 정도를 Numeric Rating Scale로 평가하였고17, Okada의 연구에서는 visual analog scale로 평가하였다21.
(5) 이상반응
이상반응은 총4개 논문에서 평가되었다5,18,19,22. 이중 National Cancer Institute Common Terminology Criteria for Adverse Events(CTCAE) 기준을 사용한 것은 3편이었다18,19,22.
(6) 기 타
Hoshino의 연구에서는 수술후 영양상태 및 대사체 분석에대해 언급하였다17.
6) 위장관운동성개선효과(Table 6)
Table 6
The Results and Conclusion in the Aspect of Intestinal Motility Improvement
| Study | Results | Conclusion |
|---|---|---|
| Hoshino, 201717 | Not available (protocol paper) | Not available (protocol paper) |
|
|
||
| Yamada, 201715 |
- The two groups did not significantly differ in the total number of residual markers (Day 1; P=0.27, Day 3: P=0.10, Day 5: P=0.09) or numbers of residual markers in the small intestine (Day 1: P= 0.36, Day 3: P=0.46, Day 5: P=0.34). - The total number of residual markers in the DKT group tended to be less than that in the no-DKT group on PODs 3 and 5. - Among the patients ≥75 years of age, the total number of residual markers in the DKT group was significantly less than in the no-DKT group on POD 5 but not PODs 1 or 3 (P=0.049). |
Although DKT had some small effect on the postoperative intestinal motility for most patients, it may have positive effects in elderly patients. |
|
|
||
| Okada, 201621 |
- Postoperative paralytic ileus was met in 35 patients (33.7%) in the TJ-100 group and 38 patients (36.9%) in the placebo group (risk difference (%), 3.2; 97.5% CI, 11.6 to 18.1; P=0.626). - The time to first flatus was median 2.25 (2.00-0.50) days in the TJ-100 group and 2.50 (1.50-0.50) days in the placebo group (relative risk, 1.12; 97.5% CI, 0.82-1.53; P=0.343). |
Use of TJ-100 did not improve recovery from paralytic ileus after PD. |
|
|
||
| Katsuno, 20164 |
- Total transit analysis: the number of markers in the DKT group (1.49±4.19) and the placebo group (1.28±3.06). There was no significant difference between the two groups (p=0.584). - Segmental transit analysis: the number of markers distributed in the small intestine or between the small intestine and the anus at 6 h was significantly greater in the DKT group than in the placebo group (15.19 vs 10.06, p=0.008). - Time to first flatus: the mean time to first postoperative flatus was 52.6±30.3 h in the DKT group and 55.6±58.6 h in the placebo group. There was no significant difference between the two groups. |
Daikenchuto has a positive effect on the resolution of delayed gastric emptying, but has a limited effect on the resolution of postoperative paralytic ileus after open surgery in patients with sigmoid or rectosigmoid cancer. |
|
|
||
| Yoshikawa, 201519 |
- Median time from extubation until first flatus was 68.9 hours (range 9.1 to 234.5 hours) in the DKT group and 68.3 hours (range 1.1 to 259.8 hours) in the placebo group (p=0.95). - Median time from extubation until first defecation was 94.7 hours (range 22.8 to 257.5 hours) in the DKT group, and 113.9 hours (range 27.7 to 284.3 hours) in the placebo group (p=0.05). - The incidence of bowel movement disorder was significantly lower in the DKT group at POD 12 (p=0.05). - There was no significant difference between the 2 groups in the presence or absence of postoperative ileus. |
Administration of DKT during the immediate postoperative period after total gastrectomy appears to promote early recovery of postoperative bowel function. |
|
|
||
| Katsuno, 201518 |
- The time of first BM: no significant difference was observed between the two groups. - Stool forms: there was no significant difference in between the two groups. - The average number of BMs: for the DKT and placebo groups on POD2 was 0.8 and 0.6, respectively. The number of BMs for the DKT group on POD6 increased to 2.2, decreasing however to 1.9 on POD8. In the placebo group, the number of BMs increased up to 2.4 on POD8. - The frequency of BM in the DKT group at POD8 was significantly lower than that in the placebo group (Wilcoxon’s rank-sum test, P=0.024). |
Although Daikenchuto had an effect on gastrointestinal function after open surgery in patients with colon cancer, this study did not show its clinical benefits adequately. |
|
|
||
| Akamaru, 20155 |
- The mean time before the first bowel movement after surgery was 4.9±2.5 days in the DKT group and 4.7±2.2 days in the control group (P=0.811). - The number of stools per day was 1.1±0.6 in the DKT group and 0.8±0.4 in the control group (P=0.037). - Stool consistency: 3.7±0.8 points in the DKT and 3.1±0.8 points in the control group (P=0.041). - Gas volume score: treatment group showed a significant decrease in intestinal gas volume compared to the control group at 7 days (78±25 vs 108±35%; P<0.05), at 1 month (70±26 vs 95±49%; P<0.05), and at 3 months (62±33 vs 90±38%; P<0.05) after surgery. |
DKT improved bowel movements, stool properties, and bowel gas. These results suggested that DKT promoted early postoperative bowel functions after total gastrectomy. |
|
|
||
| Shimada, 201522 | - The time from extubation until the first postoperative bowel movement: 88.2 h (95% CI 74.0–4.1) in the TU-100 group and 93.1 h (95% CI 83.3-9.4) in the placebo group, (P=0.0467 by log-rank test; P=0.0471 by Cox regression analysis; HR=1.327). | TU-100 appears to improve gastrointestinal dysmotility with grade B liver damage after hepatectomy. |
|
|
||
| Endo, 200620 |
- Emptying study of the pouch: both liquid and solid radioactivities decreased more rapidly during the on-treatment period than during the off-treatment period (liquid, P=0.01; solid, P=0.015). - Manometric findings: oral intake of Daikenchuto appeared to increase bursts of contractions in the pouch. During quantitative measurements, the drug significantly increased the percentage of time of contractile bursts of the pouch compared with that before the drug (P=.028) |
Daikenchuto increased intestinal motility and decreased postoperative symptoms of patients with total gastrectomy with jejunal pouch interposition |
7) 이상반응 결과
총 6편4,5,18,19,21,22의 연구에서 이상반응에 대해 보고하였다. Katsuno4의 연구에서 Common Terminology Criteria for Adverse Events(CTCAE)기준으로 3등급의 설사가 위약군에서 2건 발생하였으나, 대건중탕군과 위약군 간의 이상반응 발생빈도에 차이는 없었다. 4등급의 이상반응은 두 군에서 모두 보고되지 않았다. Katsuno18의 연구에서 3등급의 이상반응은 시험군과 대조군에서 각각 7건이 발생하였으나 발생빈도는 군간 차이가 없었으며, 4등급의 이상반응은 양군에서 모두 발생하지 않았다. Yoshikawa19의 연구에서도 3등급 이상의 이상반응 발생에 대하여 기술하였다. 대건중탕군에서는 설사 2건, 식욕부진 1건, 저알부민혈증 1건, 오심 1건, 아밀라아제 수치 상승 1건이 발생하여 총 6건의 이상반응이 보고되었다. 위약군에서는 설사 1건, 연하곤란 1건, 오심 1건으로 총 3건이 보고되었다. 그러나 두 군간의 이상반응 발생빈도에는 유의한 차이가 없었으며, 이상반응과 시험약과의 인과관계도 존재하지 않는다고 기술하였다. Okada21의 연구에서는 수술 이후 위약군에서 사망이 1건 발생하였으며, 3등급과 4등급의 이상반응은 대건중탕군에서 12명, 위약군에서 8명이 발생하였다. 이상반응의 주요 내용은 설사 혹은 혈액검사 수치결과의 이상이었다. Shimada22의 연구에서는 대건중탕군에서 3등급의 이상반응으로 고빌리루빈혈증 1건, 마비성 장폐쇄 1건, 설사 1건이 발생했고, 5등급의 이상반응으로 상부위장관 출혈 1건이 발생했다고 보고하였다. 위약군에서는 3등급 혹은 4등급의 이상반응으로 복통 3건, 오심 1건, 구토 1건이 발생했으며, 5등급의 이상반응으로 복부 출혈 1건이 발생했다고 보고하였다. 양 군간의 3등급 이상반응 발생빈도의 유의한 차이는 없었으며, 이상반응과 시험약과의 인과관계도 존재하지 않는다고 기술하였다. Akamaru5의 연구에서는 대건중탕과 관련이 있다고 의심되는 이상반응은 보고된 바가 없다고 간략하게 기술하였다. 프로토콜 논문을 제외한 나머지 2편의 연구에서는15,20 이상반응에 대한 언급이 없었다.
3. 연구의 비뚤림 위험 평가
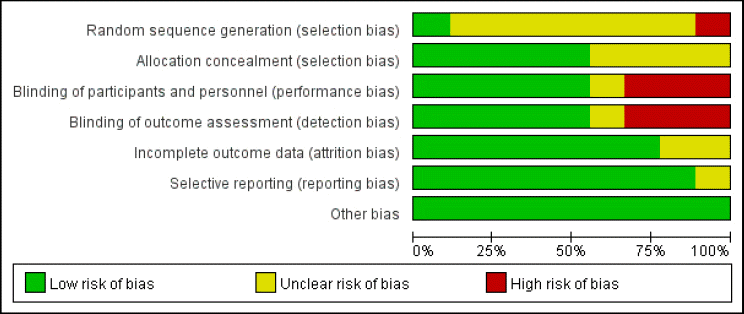
선정된 9편의 논문을 Risk of Bias16 평가방법을 적용하여 연구의 질을 평가하였다. 전체적으로 ‘Low risk of bias’가 전체항목의 63.5%로 비뚤림 정도가 높지 않았다. ‘Random sequence generation’ 항목을 보면, Hoshino17의 연구에서는 컴퓨터로 생성한 난수표를 사용했다고 기술되어있어 ‘Low’로 평가하였다. 한편, Okada21의 연구에서는 primary disease와 수술전 여부, 기관 등을 고려하여 무작위배정을 한 것으로 미루어볼 때, 임상의의 판단이 개입되었다고 생각하여 ‘High’로 평가하였다. Yamada의 연구는15 비무작위 후향적 연구로 이 항목에 해당되지 않기에 ‘Unclear’로 평가하였다. 그 외 모든 연구는 무작위 배정의 구체적 방법에 대한 언급이 없어 ‘Unclear’로 평가하였다. ‘Allocation concealment’ 항목의 경우 5편의 연구가 Central allocation을 시행하여 ‘Low’로 평가하였으며4,17-19,21, 나머지 연구는 모두 ‘Unclear’로 평가하였다. ‘Blinding of participants’의 경우 4편의 연구가 이중맹검을 시행하여 ‘Low’로 평가하였고4,18,19,21, Yamada의 연구는 맹검에 대한 언급이 없지만 모두 객관적 평가변수를 사용하여 ‘Low’로 평가하였다15. 3편의 연구는 맹검을 시행하지 않았으나 주관적 평가변수가 포함되어 있어 비맹검이 평가에 영향을 줄 수 있다고 판단하여 ‘High’로 평가하였다5,17,20. ‘Blinding of outcome assessment’의 경우에도 평가자 맹검영역과 동일한 판단 근거 및 결과가 적용되었다. 단, Yamada의 연구의 경우 ‘평가자 맹검’이라는 기술이 있어 ‘Low’로 평가하였다15. ‘Incomplete data’ 항목의 경우, 정확히 언급되지 않은 2개의 연구15,17를 제외하고는 잘 기술되어 있어 모두 ‘low’로 평가되었다. ‘Selective reporting’ 항목에서는 결과가 기재되지 않은 protocol 논문인 Hoshino의 연구17를 제외하고는 모두 ‘low’로 평가되었다. 그 외 ‘other bias’ 항목은 ‘Low’로 평가하였다(Fig. 2, 3).
III. 고 찰
실제 한의임상현장에서 암환자의 위장관 수술이후 저하된 위장관 운동성을 개선하기 위해 대건중탕을 다빈도로 사용한다. 본 연구는 이에 대한 근거를 마련하는 차원에서 시행되었다. 이에 2000년 이후에 수행된, 위장관 수술이후에 대건중탕을 투여한 임상연구 논문들을 분석, 비교하였다. 선정된 총 9편의 논문이 모두 일본에서 이루어진 연구였으며, 한국에서 수행된 임상연구는 아쉽게도 찾아볼 수 없었다. 대건중탕이라는 약의 이름도 논문에는 ‘Daikenchuto’로 표기되어 Kampo medicine으로 세계에 알려지고 있는 상황이다. 이를 볼 때, 한국인을 대상으로한 대건중탕에 대한 한의학 연구가 필요함을 확인할 수 있었다.
대건중탕의 효과를 살펴보면, 결과를 기술한 8편의 논문 중 6편이 대건중탕이 위장관 운동성을 개선함을 시사하였다. 기존 연구에 따르면, 대건중탕은 장혈류를 개선하고 점막손상을 보호함으로서 대장활성도를 조절한다고 보고되었다23,24. 또 다른 연구에서는, 대건중탕이 주로 칼시토닌 유전자 관련 단백질과 그 수용체 성분에 의해 매개되는 대장혈관전도성을 증가시킴을 발견하였다13. 위장의 운동성 관련한 연구도 찾아볼 수 있었는데, Mochiki 등은 대건중탕이 개의유문, 십이지장, 공장에서 위상성수축을 유발함을 확인하였다25. 또한 대건중탕이 위강내에서 장내뉴런을 자극하여 신경전달물질인 아세틸콜린을 분비하게 한다는 보고도 있었다26. 이러한 기전을 통해 대건중탕이 위장관 운동성을 개선한 것으로 사료된다. 한편, 이 8편의 논문을 Risk of Bias 방법을 이용하여 평가한 결과는 ‘Low risk of bias’가 전체항목의 66%로 비뚤림 정도가 높지 않았다. 이를 볼 때, 위장관 운동성 개선에 대한 결과도 신뢰할 만하다고 볼 수 있을 것이다.
효과를 보이지 않은 연구 2편 중 Okada의 연구에서는 대건중탕이 운동성 평가지표에서 위약군에비해 유의한 차이를 나타내지 않았다. 저자는 이러한 결과가 외과수술의 발전으로 인해 수술후 첫 가스배출까지의 시간이 짧아진 것으로부터 기인한 것이라고 언급하고 있다. 더불어, 수술이후에 무조건적으로 대건중탕을 투여하는 것은 지양해야한다고 결론을 내리고 있다. 이는 Kampo의학에서 한약 투여가 대증요법식으로 이루어지고 있음을 반영하는 것으로, 한의학에서 환자의 개별상태 혹은 변증에 따라 한약을 가감하여 처방하는 것이 필요하다. 일괄적인 처방이 아닌 개인 맞춤 처방이 이루어졌다면 또 다른 결과를 기대할 수 있으리라 사료된다.
또한 본 문헌고찰의 목적은 대건중탕의 효과를 확인하는 것 뿐만 아니라 추후연구를 위한 임상연구설계를 탐색하는 것도 있었다. 따라서 계획에 대해서 기술한 임상시험 프로토콜 논문도 최종 논문으로 선정하였으며, 임상연구설계의 기본적인 요소들을 분석하였다.
기존의 ‘천연물 신약⋅한약제제 임상시험 가이드라인 연구’에서는 ‘임상시험은 무작위의 대조시험설계를 위주로 하고, 이중맹검을 원칙으로 한다’는 실시원칙을 제시하고 있다. 이는 추후 임상시험설계에서도 고려되어야 할 것이다. 9편의 연구 중 이중맹검을 시행한 연구가 4편, 무작위 배정연구 8편으로 전반적인 임상연구의 기본설계가 잘 되어있었다. 단, 이중맹검을 시행하지 않은 연구에서는 평가자 맹검에 대한 언급이 없었다. 추후 임상연구설계시 위약을 사용한 이중맹검이 불가능한 경우라면 평가자 맹검을 고려해야 할 것이다.
한편, 모든 연구가 수술후에 상태를 개선하기 위한 목적으로 대건중탕을 투여하는 것이어서, 임상 연구의 과정에 다양한 수술이 포함되어 있었고, 수술직후에 대건중탕 투여가 시행되었다. 이러한 설계가 국내의 이원화된 의료체계에서 적용되려면 양한방의 협진이 잘 이루어져야 할 것이다. 현실적으로는, 암환자가 수술후 위장관 기능저하 치료를 위해 한방병원에 내원하는 경우, 수술을 시행하고 시간이 꽤 경과한 후일 경우가 대부분이다. 이 경우 약의 효과가 제대로 발현되지 않을 수 있다.
투약기간의 경우 5일에서 12주까지 다양했는데, 5일과 같이 상대적으로 짧은 기간 동안 투여한 것은 한약복용을 수술직후의 기간으로 한정시켰기 때문으로 판단된다. 추후 임상 연구 설계시에는 연구의 목적에 부합하면서 약의 효과가 충분히 나타날 수 있도록 투여기간을 산정하는 것이 필요할 것이다.
연구대상자 대상 질환의 경우 대장암이 4편으로 가장 많았으며, 그다음으로 위암이 3편이었는데 위암으로 인한 수술은 모두 전절제를 시행하였다. 그 외 췌장암, 간암관련 연구가 1편씩이었다. 우리나라에서 유병률이 높은 위암과 대장암에 대해서 연구할 때 참고할 수 있겠다. 병기는 주로 1-3기를 대상으로 하였으며, 환자상태에 대해 ECOG를 적용한 논문은 8편으로, 임상 연구 선정 기준을 정할 때 병기와 performance status를 구체화하는 것은 적용해 볼 수 있는 항목이다.
시험약은 모두 대건중탕으로 대부분의 연구에서 GMP(Good Manufacturing Practice) 시설이 갖춰진 제약회사에서 생산되었다. 추후 임상연구용 한약을 마련하는 기준으로 삼을 수 있겠으나, 현실적으로 개별연구자에게 일괄적으로 적용하기에는 어려움이 있다. 오랜 기간 동안 투여해 왔고 그 효과와 안전성이 경험적으로 입증된 한약에 대해서 좀 더 현실적인 제도 마련이 필요하다고 본다.
한편, 연구대상자의 변증유형에 따른 개인 맞춤형(individualized) 한약을 사용한 연구는 없었다. 이는 일괄 생산된 한약을 사용한 임상 환경상 피할 수 없는 상황일 수는 있겠으나, 한의학적 치료는 환자의 여러 증상을 종합적으로 살핀 후 치료를 결정하는 변증(辨證)의 과정을 거치는 것이 기본이다. 이 부분은 아쉬움이 남는다. 유의한 효과가 나타나지 않았던 연구도, 개별 맞춤형 한약을 사용했다면 또 다른 결과를 도출했을 가능성은 충분해 보인다. 만약 변증을 고려한다면 변증에 맞춘 한약제제의 마련과 변증에 대한 교육 및 연구자간의 진단일치도 제고가 이루어져야할 것이다.
대조약은 주로 위약이 대조군으로 쓰였다. 위약도 시험약과 마찬가지로 GMP 시설을 갖춘 제약회사에서 생산된 것이었다. 그 외에 휴약기를 가지는 군을 대조군으로 두는 교차설계 연구가 있었다. 국내 임상시험 가이드라인에 ‘위약대조군시험은 해당 생약(한약)제제를 이용한 치료법 자체의 가치가 있는지 없는지의 여부를 알기 위해서라기보다는 그치료법이 대조군으로 설정된 치료법 보다 가치가 뛰어난지 아닌지의 여부를 판명하기 위해 수행한다.’는 내용이 있다. 기존 표준치료를 시행하지 않는 것이 윤리적으로 문제가 되지 않을 경우에 위약을 고려해 볼 수 있을 것이다.
유효성 평가지표로는 주관적 및 객관적 지표가 다양하게 제시되었다. 주관적 지표로는 4편의 논문에서 삶의 질을 평가하였다. 암환자의 삶의 질 평가는 생존율 혹은 종양크기의 축소 외에도 암환자 치료 및 평가에서 중요한 사안으로 인식되고 있으며27, 관련된 연구가 많이 수행되고 있다28. 현재 암종별로 특이적인 설문지 연구가 많이 되어있는 상황이다. 이를 고려할 때, 특이적 삶의 질 평가도구를 장착한 임상시험을 수행한다면 의미가 있을 것으로 사료된다. 고찰대상논문 중에서도 2편의 연구가 각각 위암과 대장암 특이 삶의 질 평가도구(FACT-Ga, FACT-C)를 사용하였다.
객관적 지표로는 위장관 운동성을 평가하는 위배출능 및 운동성 측정지표, 혈액학적 검사 등을 사용했다. 위장관 운동성을 평가는 지표는 매우 다양했는데, 수술후 1일 장운동횟수, 수술후 첫 가스배출까지의 시간, 수술후 첫 장관운동까지의 시간, 수술후 마비성장폐색증 발생률 및 1일 배변횟수, 배변양상 등을 여러 편의 논문에서 확인하였고, 그 외에는 수술후 첫 배변까지의 시간, 수술후 마비성장폐색증 발생까지의 시간, 수술후 심각한 장관운동이상 존재여부, 장관내 가스양 등이 있었다. 특이한 지표로 수술후 장관 운동성 평가시 radiopaque marker를 transit analysis 연구가 2편 있었다. Transit analysis는 간편하여 수술직후 환자에게 적용하기 좋다는 장점을 가지고 있다4. 이외에 manometric study와 scintigraphic study를 이용한 연구도 있었다. 이와 같은 객관적인 지표를 이용한다면 효과 평가에 있어 연구의 질을 높일 수 있을 것이다. 현실적인 수행 가능성을 고려하여 임상연구목적에 맞는 평가지표를 적절히 활용해야할 것이다.
안전성 평가는 이상반응 파악위주로 이루어졌다. 기존 가이드라인에서 권고하는 바와 같이 CTCAE의 국제적인 기준에 의해 분석한 연구는 9편 중 5개로 상대적으로 적은 편이었고, 1편의 연구는 평가기준에 대한 언급이 특별히 없었다. 이상반응 분석을 통한 안전성 평가 결과, CTCAE 기준 3등급 이상의 이상반응이 발생한 연구가 5편 있었으나, 대건중탕군과 대조군간 발생 빈도에 유의한 차이가 없었고, 이상반응이 시험약과 관련이 있다고 보고한 연구는 없었다. 실제 임상 연구시 안전성 평가는 다각적 방법으로 자료를 얻어 수행되는 것이 적합하며, 확인된 이상반응은 투약기간, 회복시간, 연령, 용량 또는 혈중농도 등과 관련하여 지속 관찰해야 한다. 또한, 필요시 유관기관에 보고해야하며 이를 지침에 명기해야할 것이다.
본 연구에서는 소화기암 환자의 수술 후 위장관 운동성 개선을 위한 대건중탕의 효과를 탐색한 기존 임상 연구를 분석했다. 실제 임상현장에서 많은 암환자들이 수술, 항암화학요법, 방사선요법으로 인한 부작용을 개선하려는 목적으로 한의의료기관에 내원한다. 이때 환자들이 한의치료에 대한 근거를 요구하는 경우가 많은데, 본 연구에서 고찰한 논문들은 수술로 인한 부작용 개선에 한약을 사용한 근거로 유용하게 활용될 수 있다. 이상의 고찰을 통해 대건중탕의 임상적용의 근거를 마련하였으며, 나아가 국내 임상 연구설계를 위한 기반으로 사용될 수 있을 것이다.
IV. 결 론
Pubmed와 CNKI 및 국내 논문 데이터베이스 검색을 통해 선별된 소화기암 수술로 인한 위장관 운동성 개선에 대한 대건중탕에 대한 임상연구 9편을 분석하여 다음과 같은 결과를 얻었다.
1. 연구방법은 대부분 전향적 무작위 배정 임상시험으로 진행되었고, 그 외 후향적 연구도 존재하였다. 이중맹검을 시행한 연구는 4편이었다.
2. 연구대상자 대상 질환의 경우 대장암이 4편으로 가장 많았으며, 그다음으로 위암이 3편. 그 외 췌장암, 간암 관련 연구가 1편씩이었다.
3. 시험약은 모두 대건중탕으로 대부분의 연구에서 GMP(Good Manufacturing Practice) 시설이 갖춰진 제약회사에서 생산되었으며, 투약기간의 경우 5일에서 12주까지 다양했다.
4. 대조약은 주로 위약이 대조군으로 쓰였으며, GMP 시설을 갖춘 제약회사에서 생산된 것이었다. 그 외에 휴약기를 가지는 군을 대조군으로 두는 교차 설계 연구가 1편 존재하였다.
5. 유효성 평가지표로 위장관 운동성을 평가하는 위배출능 및 운동성 측정지표, 혈액학적 검사, 삶의 질 등 주관적 및 객관적 지표가 다양하게 제시되었다.
6. 결과를 기술한 8편의 논문 중 6편이 대건중탕이 위장관 운동성을 개선함을 시사하였다.
7. 이상반응 분석을 통한 안전성 평가 결과, CTCAE 기준 3등급 이상의 이상반응이 발생한 연구가 5편 있었으나, 대건중탕군과 대조군간 발생빈도에 유의한 차이가 없었고, 이상반응이 시험약과 관련이 있다고 보고한 연구는 없었다.
8. Risk of Bias 방법을 이용하여 평가한 질평가 결과는 ‘Low risk of bias’가 전체항목의 63.5%로 비뚤림 정도가 높지 않았다.